What is stain-intolerance?
The most common side effect of statins is a serious condition that affects almost 20% of statin users called myositis (injury to skeletal muscles), which causes severe, and in many cases, debilitating pain in muscles. This in turn leads to a kidney failure, a condition called “rhabdomyolysis.” These symptoms stop when the statin drug is stopped and come back when the drug is started again. This is “statin-intolerance.” Because of high levels of cholesterol, these patients are in urgent need of alternate agents to effectively reduce the high levels. Hundreds of doctors have used PreLipid + as a viable option along with other therapies like fish-oil, nicotinic acid, etc., for patients with statin-intolerance.
Microencapsulated phytosterol -- 100% fat-free. 100% disepersible
Phytosterols available in the market are completely insoluble in water and cannot be absorbed. This problem is overcome with a unique delivery system of microencapsulation, which allows for phytosterol in PreLipid+ to be dispersed and absorbed to provide maximum benefit. Also, unlike phytosterol esters, microencapsulated phytosterol is 100% fat-free and approved by the European Union.
PreLipid + is the ONLY product in the US with microencapsulated phytosterol.
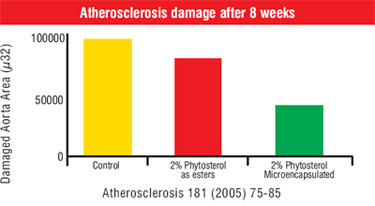
Clinical studies on Phytosterols:
More than 50 years of research and studies have shown that plant sterols lower total and LDL cholesterol levels. Two recent studies in the US:
Randomized, double-blind, 3-group parallel, controlled study from the Chicago Center for Clinical Research. Reduction in levels were total cholesterol by 6.6%, LDL by 8.1%, Apolipoprotein B by 8.4% 1 and total to HDL ratio by 8.1%. (P < 0.001 for all)
The Cardiology Department at Washington University in St. Louis with grant support from NIH studied patients who were already eating a heart-healthy diet and taking statin drugs. The addition of 2 twice-daily plant sterol in a pill form further reduced LDL cholesterol by 10%
References:
1. American Journal of Clinical Nutrition, Vol. 74, No.1, 33-43, July 2001.
2.American Journal of Cardiology, Vol. 97:3, 376-379, Feb. 2006. |

